Conversion Reactions (III) Problem 5
Description:
Problem
What reactions will bring about the following conversions?
1. 1 − Bromopropane to 2 − Bromopropane
2. Benzyl alcohol to 2 − phenylethanoic acid
3. Aniline to chlorobenzene
4. 2 − Chloropropane to 1 − propanol
Solution
1 − Bromopropane to 2 − Bromopropane
CH3CH2CH2Br → CH3 − CH(Br) − CH3
The reactant has bromine at C1 carbon while the product has bromine at C2 carbon.
Dehydrohalogenation of 1 − Bromopropane gives propene.
Markovnikov addition of HBr on propene gives 2 − Bromopropane where bromine is at C2 carbon.
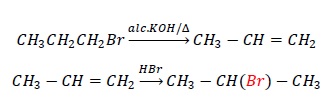
2. Benzyl alcohol to 2 − phenylethanoic acid
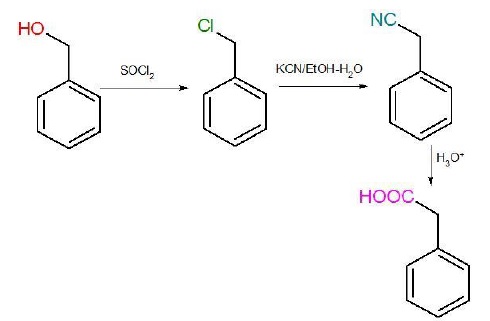
The product has 1C atom more than the reactant.
Thus, we need to convert –OH to –CN using KCN, followed by complete hydrolysis to give acid.
However, we know that direct substitution of alcohol cannot take place as –OH is a poor leaving group.
Hence, we convert it to benzyl chloride followed by nucleophilic substitution using KCN.
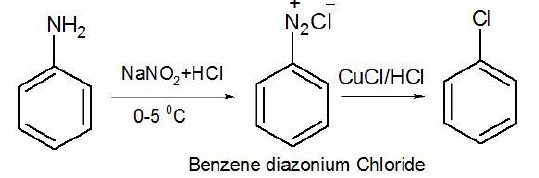
3. Aniline to chlorobenzene
Direct substitution of −NH2 by chlorine cannot take place as NH2 is a very poor leaving group.
Thus, it is first converted to diazonium salt followed by substitution with CuCl/HCl.
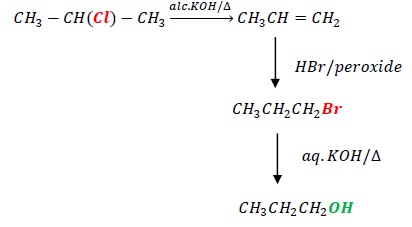
4. 2 − Chloropropane to 1 − propanol
CH3CH(Cl)CH3 → CH3CH2CH2OH
The reactant has –Cl at C2 while product has –OH at C1.
First, convert it into an alkene followed by Antimarkovnikov addition of HBr.
Lastly, substitution with aqueous KOH is carried out to get 1 − propanol as the final product.
The steps involved are dehydrohalogenation, using alc.KOH/Δ, reaction with HBr/peroxide to get Antimarkovnikov’s product CH3CH2CH2Br, substitution using aq.KOH/Δ.