Problem on Reactions of Haloarenes
Description:
Problem
Match the reactions of haloarenes given in column (A) to the type of reactions given in column (B).
| (A) | (B) |
|---|---|
(i) 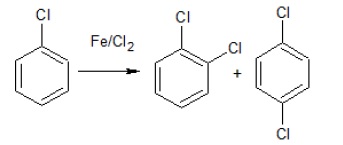 | (a) Nucleophilic Aromatic Substitution Reaction |
(ii) 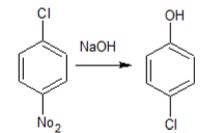 | (b) Wurtz-Fittig Reaction |
(iii) 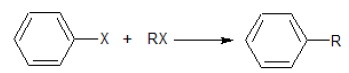 | (c) Sandmeyer Reaction |
(iv) 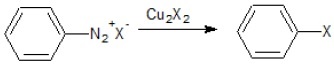 | (d) Electrophilic Aromatic Substitution Reaction |
Solution
(i) → (d), (ii) → (a), (iii) → (b), (iv) → (c)
(i) It is an example of electrophilic aromatic substitution reaction. The electrophile generated is 𝐶𝑙+ which attacks the electron rich benzene ring giving substitution at ortho − and para − positions.
(ii) This reaction is an example of nucleophilic aromatic substitution reaction as the −Cl gets substituted by –OH group. Haloarenes are generally unreactive towards this reaction, except, in the presence of electron-withdrawing groups like −NO2 when present at ortho and/or para positions. The negative charge on the carbon atom at ortho and para position is withdrawn by −NO2 group and thus stabilizes it.
(iii) The formation of alkyl arene from the reaction of haloarene and haloalkane (in the presence of Na) is called Wurtz-Fitting reaction.
(iv) This reaction is Sandmeyer reaction where N2+X− of the diazonium salt gets substituted by −Cl when reacted with CuCl/HCl.

