Reactions of Haloalkanes - Stereochemistry (II)
Description:
Retention of Configuration − It is the retention of spatial arrangement of groups of an asymmetric center during a chemical reaction.
When two species having same relative configuration can be correlated as XCabc → YCabc, there is retention of configuration.
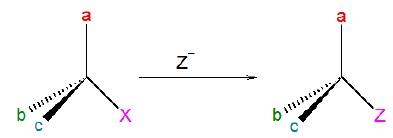
Condition for retention of configuration: No bond to the stereocenter is broken.
For example: when 2 − methylbutan − 1 − ol is heated with conc. HCl.
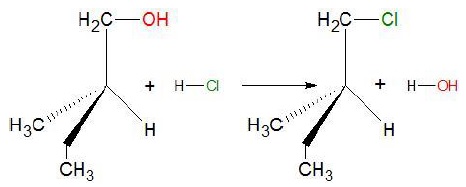
In the above reaction, we can see that no bond to the stereocenter or the asymmetric carbon atom is broken such that the configuration is retained.
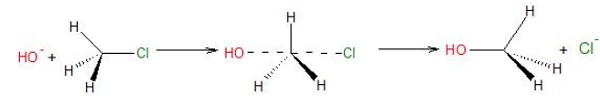
Inversion of Configuration − It is the loss of spatial arrangement of groups of an asymmetric center during a chemical reaction.
SN2 reactions involve inversion of configuration. As the nucleophile attacks the C − X bond from back side, the configuration gets inverted like an umbrella flipping upside down.
Racemisation
SN1 Mechanism
Step 1 − (CH3)3C − X ⇄ (CH3)3C+ + X−
Step 2 − OH− + (CH3)3C+ → (CH3)3C − OH
The intermediate carbocation generated is planar.
Nucleophile can attack from above or below the plane such that products are obtained with both retention and inversion of configuration.
If 50% products has inversion of configuration and remaining 50% has retention of configuration, then the resulting mixture is a racemic mixture.
SN1 Mechanism
Example
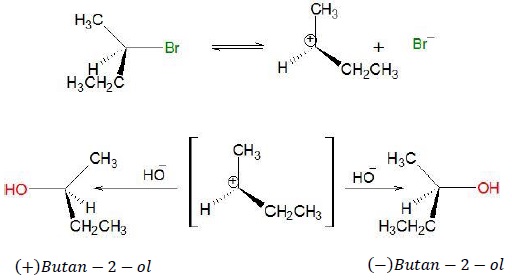
As can be seen from the mechanism, (+)Butan − 2 − ol has inverted configuration while (−)Butan − 2 − ol has retention of configuration with respected to the starting reactant.
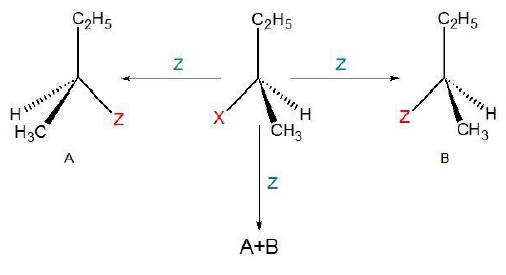
Reaction at asymmetric carbon can result in three configurations −
Retension − If only (B) is obtained.
Inversion − If only (A) is obtained.
Racemisation − If both(A) and (B) are present in 50 : 50 ratio.