Reactions of Haloalkanes - Stereochemistry (I)
Description:
Stereoisomers − are compounds of same molecular formula but differ in the way they are arranged or oriented in space.
1. Geometrical isomers: cis and trans isomers.
2. Optical isomers
Optical Isomers − Compounds (with same molecular formula) that differ in the way they rotate the plane polarized light.
Plane polarized light − when ordinary light is passed through a nicol prism or a diffraction grating such that light of a single wavelength is obtained.
The instrument used to generate plane polarized light from ordinary light is called polarimeter.
Polarimeter Set-Up: It has a source of light from which when ordinary light passes through the nicol prism, it generates plane polarized light and when this plane polarized light passes through the sample containing optically active compound, it rotates the light by some degree.
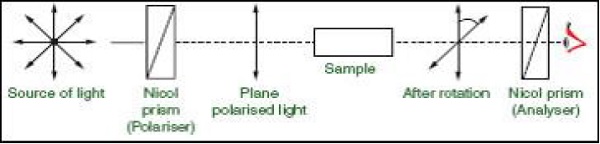
Optically active compounds can rotate the plane polarized light in
- Clockwise (dextro-rotatory, (+) d − form) or
- Anti-clockwise (laevo-rotatory, (−), l − form) direction.
- The two forms of same compound, (+) and (−) forms are called optical isomers.
Optical Activity is a very important phenomenon, the ability to rotate plane polarized light. What is the condition required for molecules to exhibit optical activity? …………
Asymmetry of the molecule!
When all 4 substituents of a carbon atom are non-identical (different), then that molecule lacks symmetry. Such a carbon atom is called asymmetric carbon atom or a stereocenter.
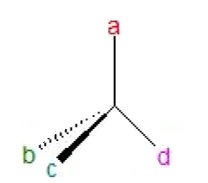
When objects have non-supersuperimposable mirror images, such an object is called chiral. For example, our hands are identical but they cannot be superimposed. However, a symmetrical object like a sphere, is identical with its mirror image as well as superimposable on it. Hence, it is an example of an achiral object.
In organic compounds, presence of an asymmetric carbon atom shows that the molecule is chiral.
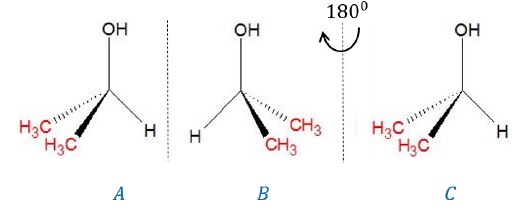
Example 1: propan-2-ol (CH3)2CH − OH
We can determine if propan-2-ol is chiral if it has an asymmetric carbon atom. However, the carbon bearing –OH group is attached to two identical methyl groups such that all 4 substituents of the carbon atom are not different. Thus, it is an achiral molecule.
We can also determine if its chiral by obtaining its mirror image, rotating it by 180o and then superimposing it. If image (C) is superimposable on (A), then it is achiral.
We can easily superimpose (C) on (A) to find that the given molecule is achiral.
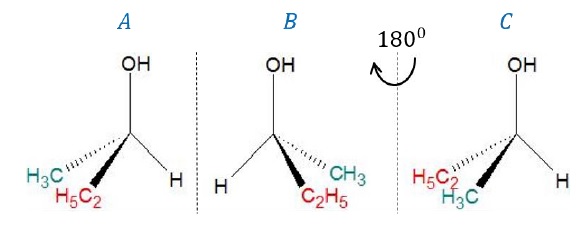
Example 2: Butan-2-ol (CH3 − CH2 − CH(OH) − CH3)
Carbon bearing the –OH group is attached to 3 different groups (−H,−CH3,−C2H5) such that it is an asymmetric carbon. Hence, the molecule is chiral.
By superimposing image (C) on (A) where (C) is obtained after rotating the mirror image (B) of butan-2-ol by 180o, we can also note that the images are not superimposable, again confirming that the molecule is chiral.
The stereoisomers which are non-superimposable mirror images of each other called enantiomers.
These possess identical physical properties like melting point, boiling point, index of refraction etc.
Only difference − rotation of plane polarised light.
When a mixture contains equal amount of both enantiomers, the net optical rotation is zero. It is because the degree of clockwise rotation caused by the d − form is cancelled exactly by anticlockwise rotation caused by l − form. This is called racemic mixture.
The process of converting an enantiomer into racemic mixture is called racemisation.

