Isomerism - Problem 4
Description:
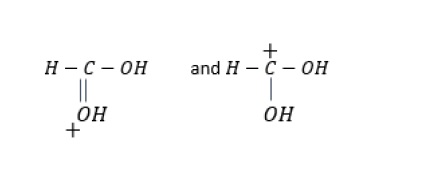
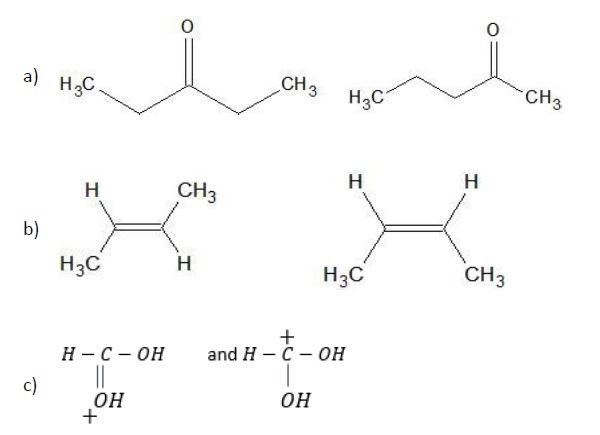
Problem − What is the relationship between the following pairs of structures?
Solution −
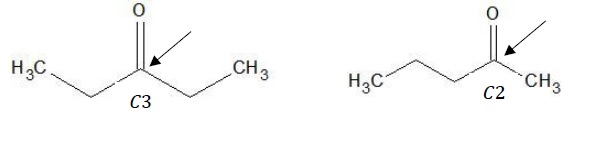
a) Position Isomerism − The position of the keto functional group has shifted from C − 3 to C − 2.
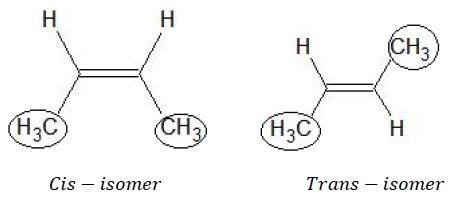
b) Geometrical Isomerism − The position of the methyl group is on the same side of the double bond giving cis-isomer while in the second structure, the methyl groups are on the opposite sides giving trans−isomer. They are thus geometrical isomers of each other.
c) Resonance structures − Both the structures are same with the exception that the positive charge has shifted from oxygen atom in the first structure to carbon in the second structure due to shifting of the π −electrons.