IUPAC Nomenclature - General Structure
Description:
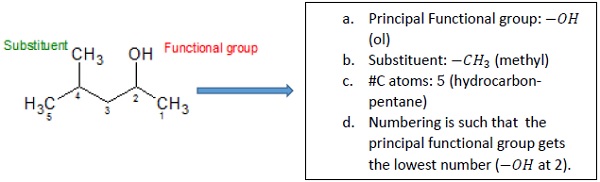
For example: The IUPAC name of the given compound is 4 − Methyl − pentan − 2 − ol
Note − Name of the functional group changes when it acts as a prefix and suffix (as given in the table below).
| Class of compounds | Functional group structure | IUPAC group-Prefix | IUPAC group-Suffix | Example |
|---|---|---|---|---|
| Alkanes | - | -ane | Butane CH3CH2CH2CH3 | |
| Alkenes | -C = C- | - | -ene | But-1-ene CH2 = CH - CH2CH3 |
| Alkynes | -C ≡ C- | - | -yne | But-1-yne HC ≡ C - CH2CH3 |
| Arenes | - | - | - | Benzene |
| Halidies | -X (X = F,Cl,Br,I) | Halo | - | 1-Bromobutane (CH3CH2CH2CH2Br) |
| Alcohols | -OH | Hydroxy | -ol | Butan-1-ol (CH3CH2CH2CH2OH) |
| Aldehydes | -CHO | Formyl or oxo | -al | Butan-1-al CH3CH2CH2CHO |
| Ketones | -C = O | oxo | -one | Butan-2-one CH3 - CO - CH2CH3 |
| Nitriles | -C ≡ N | cyano | nitrile | Butane nitrile CH3CH2CH2CN |
| Ethers | R - O - R | alkoxy | - | Ethoxy ethane C2H5 - O - C2H5 |
| Carboxylic Acids | R - COOH | - | -oic acid | Butanoic acid CH3CH2CH2COOH |
| Carboxylate ions | R - COO- | - | -oate | Sodium butanote CH3CH2CH2COO-Na+ |
| Esters | RCOOR | Alkoxycarbonyl | -oate | Methyl Propanoate CH3CH2COOCH3 |
| Actyl halides | RCOCl | Halo carbonyl | -oyl halide | Butanoyl chloride CH3CH2CH2COCl |
| Amines | RNH2 | Amino | -amine | Butanamine CH3CH2CH2CH2NH2 |
| Amides | RCONH2 | -carbamoyl | -amide | Propanamide CH3CH2CONH2 |
| Nitro Compounds | RNO2 | Nitro | - | 1-nitrobutane CH3CH2CH2CH2NO2 |
| Sulphonic acids | -SO3H | Sulpho | Sulphonic acid | Methyl sulphonic acid CH3SO3H |
Compare the two cases where OH acts as a suffix and prefix.
| Structure | Description | IUPAC Name |
|---|---|---|
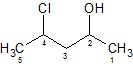 |
| 4 − Chloropentan − 2 − ol |
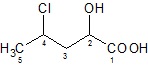 |
| 4 − Chloro − 2 − hydroxy − pentanoic acid |

