Organic Chemistry - Structural Representations
Description:
There are 4 common ways of representing structures of organic compounds.
- Dash structural formula
- Condensed structural formula
- Bond line structural formula
- 3-D structural representation
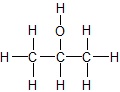
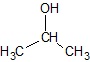
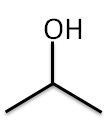
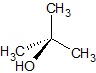
Example: C3H8O (CH3CH(OH)CH3)
Dash Structure
| Condensed Structure CH3CH(OH)CH3 (or)
| Bond line Structure
| 3-D Representation
|
Dash Structure − is obtained from Lewis dot structure of the compound, where the atoms are linked by lines.
Condensed Structure − the hydrogen atoms are written next to carbon atom.
Bond Line Structure − carbon skeleton with only heteroatoms like O,N,Cl are written.
3-D Representation − shows the placement of atoms in space.
Solid wedge represents the atom coming out of the plane or towards the viewer.

Dashed wedge represents the atom moving into the plane or away from the viewer.